En respuesta al anuncio del alcalde Ismael Burgueño Ruiz sobre...
Leer más
FDA: Moderna Vaccine Safe for Kids 6 Months to 17 Years
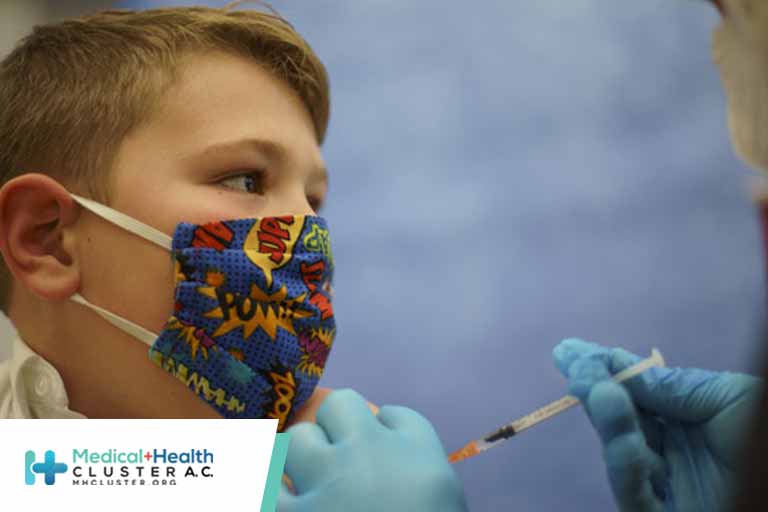
U.S. Food and Drug Administration advisers say Moderna’s COVID-19 vaccine is safe and effective for children aged 6 months to 17 years, according to an FDA document made public on Friday.
Two doses of the Moderna vaccine caused an immune response in clinical trial participants that was similar to the immune response found in young adults who took the vaccine, the document said.
Children under 11 overall were noted to have had fewer adverse reactions than adolescents, while children 6 years and under reported more fevers than adolescents and adults. A booster shot will probably be needed.
“Available data support the effectiveness of the Moderna COVID-19 Vaccine in preventing symptomatic COVID-19 in pediatric age groups from 6 months through 17 years of age,” the FDA wrote.
The document release comes days before FDA meetings that could help make COVID vaccines available for millions of young children in the United States.
The FDA’s Vaccines and Related Biological Products Advisory Committee meets Tuesday to consider amending the Moderna emergency use authorization (EUA) to include children 6 years to 17 years. On Wednesday, the committee will consider amending the EAUs for the Moderna vaccine to include children between 6 months and 5 years and the Pfizer/BioNTech vaccine to include children 6 months through 4 years.
Currently, children under 5 are not eligible for any COVID vaccines. Pfizer vaccines are available for people 5 and up, while the Modern and Johnson & Johnson vaccines are available for people 18 and up.
If the committee recommends approval, the CDC and FDA could authorize the vaccines for young children by June 17 or 18.
Earlier this week, the Biden administration said widespread vaccination of children under 5 could begin June 20 if the EUAs are granted. The White House said it had already pre-ordered 10 million doses of the Pfizer-BioNTech and Moderna mRNA vaccines for this age group.
https://www.medscape.com/viewarticle/975471?src=soc_fb_220613_mscpedt_news_mdscp_moderna&faf=1
Créditos: Comité científico Covid




