En atención a la creciente preocupación sobre la confianza en...
Leer más
Preventive Medication for COVID-19 Infection
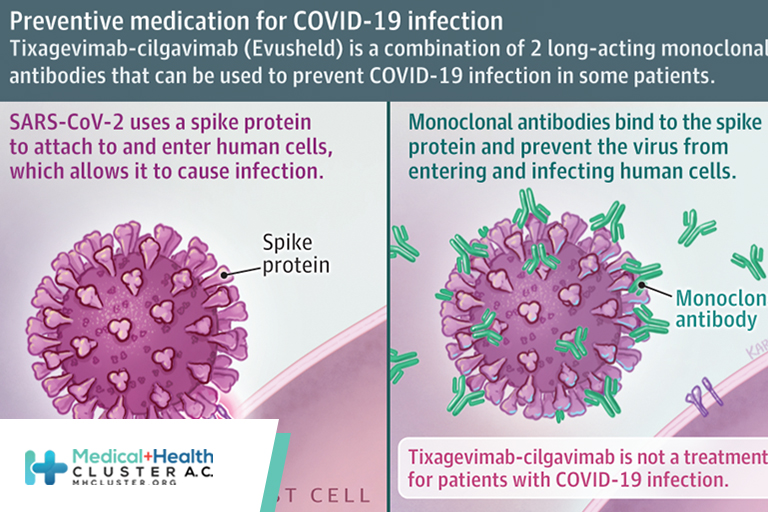
Tixagevimab-cilgavimab is a combination medication to help prevent COVID-19 infection.
Tixagevimab-cilgavimab (Evusheld) consists of 2 long-acting monoclonal antibodies that bind to the spike protein of the virus that causes COVID-19 to prevent it from infecting human cells.
When Can Tixagevimab-Cilgavimab Be Given?
Tixagevimab-cilgavimab is not a treatment for COVID-19 and should not be given to patients who are already infected with COVID-19 or who had recent close contact with an individual who has COVID-19 infection. Administration of tixagevimab-cilgavimab should be deferred for at least 2 weeks after receipt of COVID-19 vaccination.
Who Is Eligible to Receive Tixagevimab-Cilgavimab?
On December 8, 2021, the US Food and Drug Administration issued an Emergency Use Authorization (EUA) for tixagevimab-cilgavimab. Eligible patients include adults and children aged 12 years or older who weigh at least 88 lb (40 kg) and are moderately to severely immunocompromised due to a medical condition or immunosuppressive medications and may have an inadequate immune response to COVID-19 vaccination or cannot be vaccinated due to a history of severe reactions to a COVID-19 vaccine.
Where and How Is Tixagevimab-Cilgavimab Given?
Tixagevimab-cilgavimab is given by 2 separate consecutive intramuscular injections during a single session at a doctor’s office, infusion center, or other health care facility. Individuals are monitored for adverse reactions on site for at least 1 hour after receiving tixagevimab-cilgavimab. If ongoing protection against COVID-19 is needed, tixagevimab-cilgavimab is currently given every 6 months. People who meet eligibility criteria for tixagevimab-cilgavimab should contact their doctor to find out where to receive this treatment, which is provided to patients at no cost.
How Effective Is Tixagevimab-Cilgavimab?
Tixagevimab-cilgavimab has been shown to substantially decrease the risk of developing symptomatic COVID-19 infection for up to 6 months after administration. Research on the protection provided by tixagevimab-cilgavimab is ongoing, especially because mutations in the spike protein of emerging variants of the virus that causes COVID-19 could interfere with the effectiveness of tixagevimab-cilgavimab.
Possible Side Effects of Tixagevimab-Cilgavimab
Potential side effects of receiving an intramuscular medication include pain, soreness, swelling, bruising, and possible bleeding or infection of the skin at the site of injection. Allergic reactions can occur during or after administration of tixagevimab-cilgavimab and may include symptoms such as shortness of breath, chest pain, hives, wheeze, and swelling of the face, lips, mouth, and tongue. Individuals who have had a severe allergic reaction to a COVID-19 vaccine are at higher risk of having an allergic reaction to tixagevimab-cilgavimab. Rare serious cardiac adverse events have been reported in patients with underlying cardiac risk factors who received tixagevimab-cilgavimab.
What If a Patient Who Received Tixagevimab-Cilgavimab Develops COVID-19 Symptoms?
People who develop symptoms of COVID-19 infection should be tested for the virus as soon as possible. Patients who received tixagevimab-cilgavimab and then test positive for COVID-19 may be eligible for treatment so should contact their doctor to discuss the best available COVID-19 therapeutic options
Créditos: Comité científico Covid




