On April 13, the CDC and the FDA issued a joint statement recommending a pause in Johnson & Johnson vaccine administration, pending review of six reported US cases of a rare and severe type of blood clot occurring after receiving the Johnson & Johnson vaccine. To date, more than 6.8 million doses of […]
Read More
As potentially more dangerous coronavirus variants spread worldwide, scientists and clinicians have raced to discover how well the available COVID-19 vaccines protect against the mutant strains. Preliminary results from a large study of health care workers now suggest one dose of CoronaVac, a vaccine developed by a Chinese company, is […]
Read More
The FDA and CDC on Tuesday recommended that use of the Johnson & Johnson COVID-19 be paused after reports of blood clots in patients receiving the shot, the FDA and CDC announced Tuesday. In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the […]
Read More
The US, South Africa and European Union will temporarily stop the rollout of the Johnson & Johnson (J&J) Covid jab, after reports of rare blood clotting. Six cases were detected in more than 6.8 million doses of the vaccine, the US Food and Drug Administration (FDA) said. Johnson & Johnson […]
Read More
The global effort to develop safe and effective Covid-19 vaccines has yielded remarkable results, owing in part to early, decisive investments in clinical discovery through efforts such as Operation Warp Speed. These achievements highlight the payoff of stable, long-term support of basic research and immunology: our scientific community was prepared […]
Read More
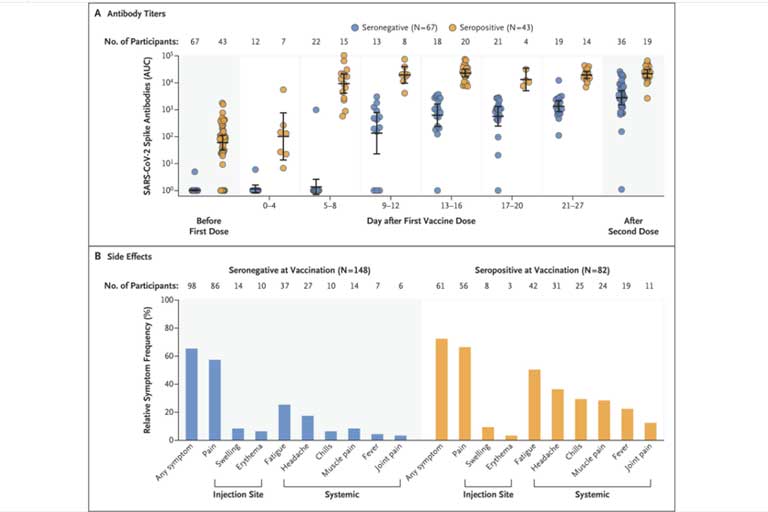
The efficacy of two injections of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike messenger RNA (mRNA) vaccines (BNT162b2 [Pfizer] and mRNA-1273 [Moderna])1 in preventing symptomatic SARS-CoV-2 infection in persons without previous coronavirus disease 2019 (Covid-19) has been shown to be high. We wondered what the response would be to the […]
Read More
The European Medicines Agency continues to reassure the public about the safety of the AstraZeneca COVID-19 vaccine, although several countries have imposed new restrictions on the product, owing to its link to a rare clotting disorder. Use of the vaccine has been suspended for individuals younger than 55 or 60 […]
Read More
A small number of people have tested positive for COVID-19 more than two weeks after being fully vaccinated, which is also known as a “breakthrough” case. The two-week mark is important because the body needs enough time to develop immunity, according to Prevention. So far, those with breakthrough cases have reported […]
Read More
Phase III clinical trials show that Pfizer’s coronavirus vaccine is 100% effective in protecting children 12-15 years old from infection, the company said in a news release. The study enrolled 2,260 adolescents ages 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European […]
Read More
Canada paused use of the AstraZeneca-University of Oxford COVID-19 vaccine on Monday for people under age 55 after concerns linked to rare blood clots, according to The Associated Press. The National Advisory Committee on Immunization, the vaccine advisory group in Canada, recommended the pause for safety reasons. “There is substantial […]
Read More